At the beginning of July, the health authorities of the United States fully authorized the use of the drug Leqembi to treat Alzheimer’s, after Be the first to demonstrate a clear reduction in cognitive decline.
The results confirmed the ability of donanemab to slow the progression of the disease in patients, especially when taken in the early stages, but some experts are wary because its effects are still “modest” and it has dangerously dangerous side effects.
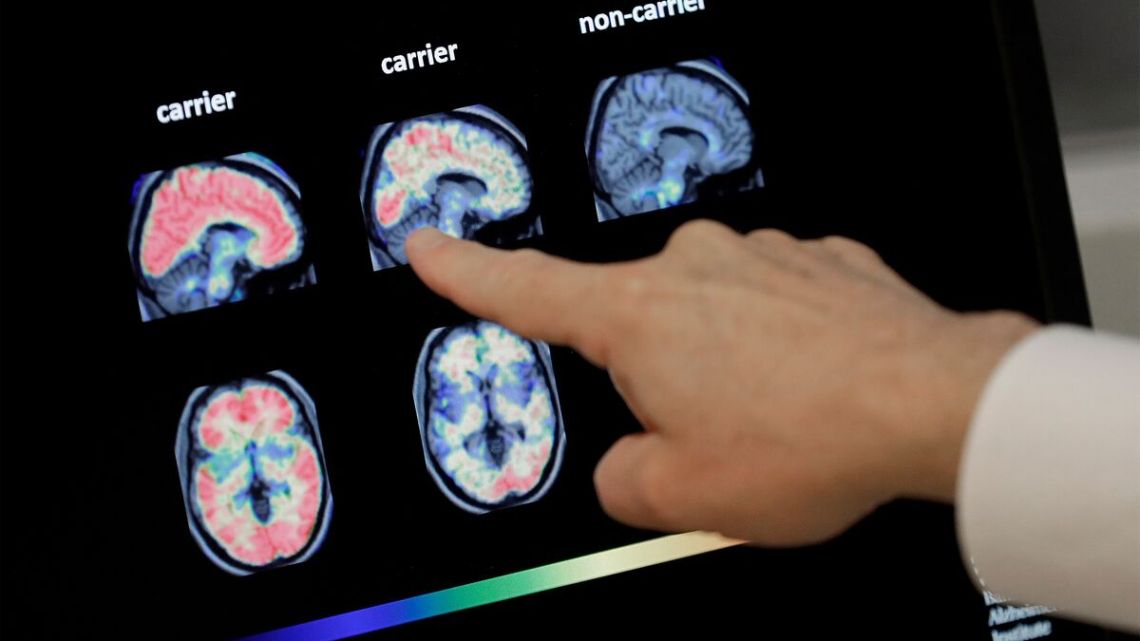
In May, the US Food and Drug Administration (FDA) came up with a similar treatment for Alzheimer’s disease, Leqembi (lecanemab), made by Eisai and Biogen. Eli Lilly said it expects US health authorities to rule “by the end of the year” and is filing applications in other countries. Donanemab, like lecanemab, is administered intravenously and attacks plaques in the brain of affected patients, known as amyloid plaques.
The clinical trial of Eli Lilly’s treatment was conducted in eight countries with more than 1,700 people between the ages of 60 and 85 who had not yet reached an advanced stage of the disease, and the results were published Monday in the scientific journal ever.
Alzheimer’s: a drug slowed the progression of the disease in 35 percent of cases
In a subgroup of about 1,200 people whose brains showed low levels of the Tau protein—indicating an even earlier stage of the disease—the treatment reduced cognitive and functional decline (ability to perform daily activities) by 35% in 18 months. But the treatment can have serious side effects, such as edema and cerebral hemorrhages.
Three deaths of participants in the clinical trial are likely related to the treatment, the study reports. “The modest benefits will probably not be questioned by patients, doctors or taxpayers” if these treatments are dangerous “low risk, cheap and simple to administer,” several experts said in a paper also published in Jama. “But none of the three things is proven.”
Collecting more data, even beyond 18 months, will be crucial to better understand the balance between the benefits and risks of these drugs, they stressed, also criticizing the low proportion of people of color included in the trial, despite the fact that They are the most affected by the disease.
These “first-generation” drugs “are not perfect,” summed up Susan Kohlhaas of the organization Alzheimer’sUK research. “But they are a big step in the right direction.” “They represent a breakthrough that will pave the way for many future treatments,” agreed Giles Hardingham, a professor of pharmacology at the University of Edinburgh.
“I got my life back”: the story of a patient who underwent a new “historical” treatment against Alzheimer’s

One of the patients who underwent clinical trials assured that she “got her life back” thanks to the drug.
Joan Murtaugh, 77, a resident of Ohio (United States), met with the tests of the drug lecanemab which were carried out at Cleveland Clinic after he was shown to have mild cognitive impairment and high risk of Alzheimer’s in 2017.
After being part of the experiments for 18 months, between 2020 and 2021, the woman was happy with the results, assuring in dialogue with Sun that it feels “perfectly normal” and that “I’m not having the problems that I have”.
Given the positive effects that the drug seems to have on his health, the septuagenarian continues to inject the drug once a week. In addition to this, it complements the treatment with cognitive speech therapy.

“I am fully functional and my memory is much better than it was. I am very active, drive regularly, garden and cook, and read several books. I got my life back, “Joan explained to the British media. In tune, her husband Larry, 77, was also satisfied with the effects of the drug, considering that”this medicine is almost a miraclefrom our perspective.”
“When it was detected in Joan, my fears were that this would be a downward spiral. It used to be that when Alzheimer’s was mentioned it was staring into an abyss. Now this drug offers great hope for the future“Added the man. About his partner, he acknowledged “seeing improvements in Joan’s short-term memory.” “She is a fighter,” he said.
According to Joan, “she had no doubts” about joining the clinical trials when she found out about the drug’s existence. “She knew that Cleveland Clinic is a first class place and when she thought they had a medicine that could help, and i need help desperately, I went for it. I had no doubts,” she explained.
Babak Tousi, a physician who led part of the trial at Cleveland Clinic, told Sun that “we have a large group of patients who they come to us and ask us for help“In particular, Joan was a citizen scientist, she asked if she could be part of a trial,” she added.
“Of course, people who participate in clinical trials may have access to drugs that are not yet available. But interestingly, a bigger factor is that they say that It may not help them, but it will help other people.. They want to return the favor,” Tousi said.
Leqembi will be “broadly” covered in the United States
In early July, US health authorities fully licensed the Alzheimer’s drug, allowing treatment is more accessible through the federal health coverage system. In that sense, Leqembi was advanced for patients who have not yet reached an advanced stage of the disease.
In January it had already been approved by the US Drug Agency (FDA) through an accelerated procedure. According to a statement released by the agency, the analysis of additional studies allowed is now authorized in its entirety.
Administered intravenously every two weeks, the treatment was developed by the Japanese pharmaceutical company Eisai with the American Biogen. Its price was set at $26,500 per year and per patient.
Alzheimer’s: 7 habits that can delay the disease for many years
The federal health coverage system Medicare, intended for people over 65, until now only covered it if it was taken in clinical trials, which greatly limited its access. With the full clearance of the FDA, it will now be “broadly” coveredhighlights Chiquita Brooks-LaSure, director of the agency that administers Medicare, in a statement.
“This is great news for the millions of people across the country living with this debilitating disease and their families,” Brooks-LaSure added. However, according to the text, patients must still pay part of the cost out of pocket (20%).
While this is not a cure for patients, can help slow down the progression of the disease a bit. “This treatment, while not a cure, can help give people more time … to maintain their independence and do the things they love,” said Joanne Pike, president of the Alzheimer’s Association. People with the disease “deserve the opportunity to discuss and decide, with their doctor and family, whether this treatment is right for them,” she added. However, it is likely to cause serious side effects, such as cerebral edema or hemorrhage, which can be fatal.
MB CP/DS